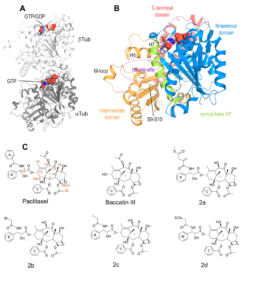
Structural insight into the stabilization 1 of microtubules by taxanes
Paclitaxel (Taxol®) is a taxane and a first-line chemotherapeutic drug that stabilizes microtubules. While the interaction of paclitaxel with microtubules is well described, the current lack of high-resolution structural information on a tubulin-taxane complex precludes a comprehensive description of the binding determinants that affect the drug’s mechanism of action. Here, we solved the crystal structure of the core baccatin III moiety of paclitaxel lacking the C13 side chain in complex with tubulin at 1.9 Å resolution. Based on this information, we engineered two tailor-made taxanes with modified C13 side chains, solved their crystal structures in complex with tubulin, and analyzed their effects along with those of paclitaxel, docetaxel, and baccatin III on the microtubule lattice by X-ray fiber diffraction. We then compared high-resolution structures of ligand-bound tubulin and microtubule complexes with apo forms and used molecular dynamics simulations to understand the consequences of taxane binding to tubulin as well as to simplified protofilament and microtubule-lattice models. Our combined approach sheds light on three mechanistic questions. Firstly, taxanes bind better to microtubules as compared to unassembled tubulin due to a dual structural mechanism: Tubulin assembly is linked to a conformational reorganization of the bM loop, which otherwise occludes ligand access to the taxane site, while the bulky C13 side chains preferentially recognize
the microtubule-assembled over the unassembled conformational state of tubulin. Second, the occupancy of the taxane site by a ligand has no influence on the straightness of tubulin protofilaments. Finally, binding of the taxane core to the taxane site displaces the S9-S10 loop of b-tubulin resulting in microtubule expansion. Our results provide
detailed new insights into the mechanism of microtubule-stabilization by taxanes.
https://www.tubintrain.eu/wp-content/uploads/2023/03/2021.07.20.453061v2.full_-1.pdf
Computational Approaches to the Rational Design of Tubulin-Targeting Agents
Microtubules are highly dynamic polymers of α,β-tubulin dimers which play an essential role in numerous cellular processes such as cell proliferation and intracellular transport, making them an attractive target for cancer and neurodegeneration research. To date, a large number of known tubulin binders were derived from natural products, while only one was developed by rational structure-based drug design. Several of these tubulin binders show promising in vitro profiles while presenting unacceptable off-target effects when tested in patients. Therefore, there is a continuing demand for the discovery of safer and more efficient tubulin-targeting agents. Since tubulin structural data is readily available, the employment of computer-aided design techniques can be a
key element to focus on the relevant chemical space and guide the design process. Due to the high diversity and quantity of structural data available, we compiled here a guide to the accessible tubulin-ligand structures. Furthermore, we review different ligand and structure-based methods recently used for the successful selection and design of new tubulin-targeting agents.
https://www.tubintrain.eu/wp-content/uploads/2023/02/biomolecules-13-00285.pdf
Maytansinol Functionalization: Towards Useful Probes for Studying Microtubule Dynamics
Maytansinoids continue to excite interest almost after a decade after their discovery as potent antimitotic tubulin binders.[1–3] Their highly efficient mechanism of capping MT-dynamics[4] has led to their successful application in cancer treatment as antibody-drug conjugates (ADCs).[5–7] This success has further prompted their development as targeted cancer therapeutics, for example, in the form of nanoparticles[8,9] or recently reported immune checkpoint-targeting maytansinoid conjugates.[10] However, the high-affinity of maytansine towards β-tubulin (KD=6.8–14 nM) not only makes it well suited as a cytotoxin, but also derivatives with a similar affinity could be used as molecular probes.[11,12] As of today, effortless generation of maytansine-based molecular probes is hampered by major drawbacks such as the complexity of the natural product scaffold and lack of SAR studies, which could suggest suitable points for attachment of fluorophore tags or radionuclides. Here, we report on both of these aspects: the chemistry of maytansinol (Figure 1, 1) for the creation of maytansinoid conjugates and the suitability of the C3-position to tolerate bulky substituents without affecting the binding mode. Therefore,
we provide a solid basis for in-depth exploration of maytansinoids as molecular probes to study microtubule dynamics. Previously, it has been shown that the ester at the C-3 position of ansamitocins, maytansine, and maytansinoids plays an important role in biological activity and cell permeability.[3,13,14] In our recent study, we extensively investigated a series of C-3 derivatized maytansinoids obtained by acylation of maytansinol.[12] By X-ray crystallography experiments, we observed that the studied maytansinoids retained a fundamental spatial arrangement with respect to β-tubulin. The binding mode of the core structure of maytansinol remained unaltered independently of the introduced substituents. Furthermore, the binding affinities of the C3-substituted maytansinoids obtained are very similar to the one of maytansine. Based on these observations, we decided to further investigate the potential of C-3 functionalized maytansinoids as molecular probes by creating novel tubulin binders. The design of novel maytansinoids was led by the high-resolution X-ray
crystallography structures of recently obtained maytansinoidtubulin complexes (PDB IDs: 5SB9, 5SBA and 5SBB). The maytansine binding site is located in proximity of the GDP nucleotide bound to the E-site and a GDP-coordinated Mg2+. The first interesting factor that could be exploited to exert a
novel effect on MTs is the Mg2+ ion coordinated by the nucleotide in close proximity to the maytansine binding site. The design of bivalent compounds containing maytansinol and nucleotide mimetics would a) increase the understanding of the ability of maytansinoids to accommodate in the binding site
independently of the size of the substituents, and b) investigate the ability of these molecules to interact with Mg2+ and to displace the nucleotide from the binding site. Tubulin inhibition by nucleotide analogues is very challenging because of GTP concentrations around 300 μM inside the cells.[15] However, we sought to investigate whether the presence of the maytansinol moiety in a bivalent compound could favor binding of the nucleotide portion through an
entropic effect. The maytansinol moiety would act as an anchor point holding the modified nucleotide portion in close proximity of the E-site through a flexible linker, thereby favoring the nucleotide exchange. Accordingly, we decided to prepare long-chain maytansinol derivatives and maytansinoid conjugates to target either the Mg2+ ion or the nucleotide binding site. Introducing different ubstituents at the 3-O position should allow the protein-ligand
interaction to be optimized and new molecules capable of producing different effects on tubulin structure and dynamics to be found
Microtubule-modulating Agents in the Fight Against Neurodegeneration: Will it ever Work?
The microtubule skeleton plays an essential role in nerve cells as the most important structural determinant of morphology and as a highway for axonal transport processes. Many neurodegenerative diseases are characterized by changes in the structure and organization of microtubules and microtubule-regulating proteins such as the microtubule-associated protein tau, which exhibits characteristic changes in a whole class of diseases collectively referred to as tauopathies. Changes in the dynamics of microtubules appear to occur early under neurodegenerative conditions and are also likely to contribute to age-related dysfunction of neurons. Thus, modulating microtubule dynamics and correcting impaired microtubule stability can be a useful neuroprotective strategy to counteract the disruption of the microtubule system in disease and aging. In this article, we review current microtubule-directed approaches for the treatment of neurodegenerative diseases with microtubules as a drug target, tau as a drug target, and post-translational modifications as potential modifiers of the microtubule system. We discuss limitations of the approaches that can be traced back to the rather unspecific mechanism of action, which causes undesirable side effects in non-neuronal cell types or which are due to the disruption of non-microtubule-related interactions. We also develop some
thoughts on how the specificity of the approaches can be improved and what further targets could be used for modulating substances.
https://www.tubintrain.eu/wp-content/uploads/2022/05/Brandt.pdf
Foldamers reveal and validate therapeutic targets associated with toxic α-synuclein self-assembly
Parkinson’s disease (PD) is a progressive neurodegenerative disorder for which there is no successful prevention or intervention. The pathological hallmark for PD involves the selfassembly of functional Alpha-Synuclein (αS) into non-functional amyloid structures. One of the potential therapeutic interventions against PD is the effective inhibition of αS aggregation. However, the bottleneck towards achieving this goal is the identification of αS domains/sequences that are essential for aggregation. Using a protein mimetic approach, we have identified αS sequences-based targets that are essential for aggregation and will have significant therapeutic implications. An extensive array of in vitro, ex vivo, and in vivo assays is utilized to validate αS sequences and their structural characteristics that are essential for aggregation and propagation of PD phenotypes. The study aids in developing significant mechanistic and therapeutic insights into various facets of αS aggregation, which will pave the way for effective treatments for PD.
Structure-based classification of tauopathies
The ordered assembly of tau protein into filaments characterizes several neurodegenerative diseases, which are called tauopathies. It was previously reported that, by cryo-electron microscopy, the structures of tau filaments from Alzheimer’s disease1,2, Pick’s disease3, chronic traumatic encephalopathy4 and corticobasal degeneration5 are distinct. Here we show that the structures of tau filaments from progressive supranuclear palsy (PSP) define a new three-layered fold. Moreover, the structures of tau filaments from globular glial tauopathy are similar to those from PSP. The tau filament fold of argyrophilic grain disease (AGD) differs, instead resembling the four-layered fold of corticobasal degeneration. The AGD fold is also observed in ageing-related tau astrogliopathy. Tau protofilament structures from inherited cases of mutations at positions +3 or +16 in intron 10 of MAPT (the microtubule-associated protein tau gene) are also identical to those from AGD, suggesting that relative overproduction of four-repeat tau can give rise to the AGD fold. Finally, the structures of tau filaments from cases of familial British dementia and familial Danish dementia are the same as those from cases of Alzheimer’s disease and primary age-related tauopathy. These findings suggest a hierarchical classification of tauopathies on the basis of their filament folds, which complements clinical diagnosis and neuropathology and also allows the identification of new entities—as we show for a case diagnosed as PSP, but with filament structures that are intermediate between those of globular glial tauopathy and PSP.
https://www.tubintrain.eu/wp-content/uploads/2021/12/17-12-2021s41586-021-03911-7.pdf
Chalcone Analogue as New Candidate for Selective Detection of α‑Synuclein Pathology
Deposition of α-synuclein (α-syn) aggregates is one of the neuropathological hallmarks of synucleinopathies including Parkinson’s disease, dementia with Lewy bodies, and multiple-system atrophy. In vivo detection of α-syn aggregates with SPECT or PET may be an effective tool for medical intervention against
synucleinopathy. In the present study, we designed and synthesized a series of chalcone analogues with different aryl groups to evaluate their potential as α-syn imaging probes. In competitive inhibition assays, aryl groups markedly affected binding affinity and selectivity for recombinant α-syn aggregates. Chalcone analogues with a 4- (dimethylamino)phenyl group bound to both α-syn and amyloid β (Aβ) aggregates while ones with a 4-nitrophenyl group displayed α- syn-selective binding. In fluorescent staining, only chalcone analogues with a 4-nitrophenyl group succeeded in selective detection of human α-syn against Aβ aggregates in patients’ brain samples. Among them, PHNP-3 exhibited the most promising binding characteristics for α-syn aggregates (Ki = 0.52 nM), encouraging us to further evaluate its utility. Then, a 125I-labeling reaction was performed to obtain [125I]PHNP-3. In a binding saturation assay, [125I]PHNP-3 bound to α-syn aggregates with high affinity (Kd = 6.9 nM) and selectivity. In a biodistribution study, [125I]PHNP-3 exhibited modest uptake (0.78% ID/g at 2 min after intravenous injection) into a normal mouse brain. Although there is room for improvement of its pharmacokinetics in the brain, encouraging in vitro results in the present study indicate that further structural optimization based on PHNP-3 might lead to the development of a clinically useful probe targeting α-syn aggregates in the future.
https://www.tubintrain.eu/wp-content/uploads/2021/12/16-12-2021-acschemneuro.1c00441.pdf